NEGATIVELY CHARGED LIPSOMES--A NEGATIVE CHARGE IS A NUCLEAR LOCALIZATION SIGNAL--STUDIES
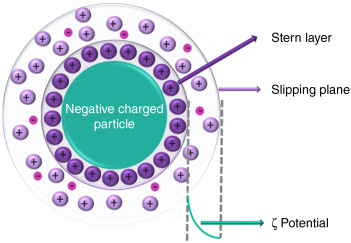
LNP is a lipid particle. The negative charge is equivalent if the zeta potential pushes negative on the LNP due to DNA contamination as the DNA is negatively charged
Nuclear gene targeting using negatively charged liposomes
Welz, C., Neuhuber, W., Schreier, H., Repp, R., Rascher, W., & Fahr, A. (2000). Nuclear gene targeting using negatively charged liposomes. International Journal of Pharmaceutics, 196(2), 251–252. doi:10.1016/s0378-5173(99)00433-0
10.1016/s0378-5173(99)00433-0
Plasmid DNA has been found by multiple researchers as a contaminant in the current covid “vaccines”.
The plasmid DNA that appears in pieces, is linearized. This means that the plasmid DNA started off as small circular pieces, but the scientists making the “vaccines’ thought they would put it through a process to “chew it up” which ended up chopping it up into more “straight line” pieces, although this does not mean they appear in a straight line, per se.
This is a very layman’s statement, of course.
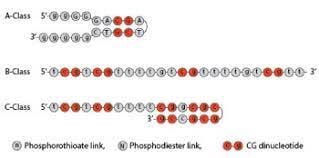
The DNA plasmid is also dsDNA, that is double stranded DNA. It contains many parts. It also contains what is called a CpG motif. Also, the pieces of plasmids, depending on size and composition, may be referred to as an oligonucleotide. This might get confusing, as we also refer to it as ODN.
Oligonucleotides are used in biotechnology, including in plasmids. Plasmids are small, circular DNA molecules found in bacteria and are used in for gene cloning, expression, and manipulation. Scientists use parts of them, and add components to them, such as antibiotic resistance genes, promoters (like the SV40), and the gene expression for proteins (and in this case, spike).
^^^ these are quick videos, but he does a good job of explaining things.
CpG motifs are sequences of DNA containing a cytosine followed by a guanine nucleotide, linked by a phosphate (CpG). These motifs are prevalent in the genome, particularly in regions called CpG islands, and they play important roles in gene regulation and immune responses. The CpG motif is what is present in the DNA plasmid used to make the “vaccines” which is present as contamination.
The Study
Experimental detail quick hit:
Liposomal Formulations Preparation:
Lipid film evaporation followed by hydration in Tris buffer.
Sonication and extrusion through 50 nm Nuclepore® membranes.
Lipid fluorescence with rhodamine-labeled DOPE.
Characteristics of AVE™-3 Liposomal Formulation:
Negatively charged.
Derived lipid composition from enveloped virus membranes.
Demonstrated fusogenic properties.
Oligonucleotide and Protamine Complex:
68-mer oligonucleotide forming intramolecular double strands.
Labeled with FAM or fluorescein.
Complexed with protamine for DNA condensation.
Experimental Procedure:
Incubation of complexes with liposomal formulations.
Transfer to HepG2 cells.
Serum addition after 3 hours if incubation exceeds 5 hours.
Nuclear Localization of Oligonucleotides:
AVE™-3 facilitates nuclear localization, especially with higher, negative charge ratios.
Neutral control liposomes do not induce nuclear uptake.
Negatively charged liposomes DO induce nuclear uptake.
Co-uptake of Negatively Charged Liposomes:
Co-localization also occurs with oligonucleotides.
Potential for targeted delivery using receptor-sensitive ligands.
More details:
Researchers in the study used oligonucleotides to control gene activity and to see if they could correct genetic diseases caused by single point mutations.
(read that again).
They used chimeric oligonucleotides.
Chimeric oligonucleotides are designed to form an intramolecular double strand of DNA and modified RNA bases, allowing them to target specific sequences within the genome.
To see what would happen to these oligonucleotides, the researchers used fluorescently labeled pure 68-mer DNA-analogue complexed with protamine sulfate and coated with three different liposomal formulations for delivery into cells.
A liposome:
What the researchers found, was that the interaction between negatively charged liposomes and protamine-complexed oligonucleotides drove the ODN into the nucleus. ODN= oligonucleotides.
The interaction between negatively charged liposomes and protamine-complexed oligonucleotides is facilitated by the negative charge.
The nuclear pore complex (that is the complex in the pores of the nucleus where our DNA is housed) contains a number of proteins with various charges.
The nuclear pore complex itself has a net negative charge, mainly due to the presence of nucleoporin proteins, which contain numerous negatively charged amino acid residues.
While the nuclear pore complex contains many negatively charged components, it also includes various proteins and structures with positive charges.
Nucleoporins, the proteins that make up the nuclear pore complex, have diverse chemical compositions, and contain positively charged amino acid residues.
Interaction with liposomes stabilize the protamine-complexed oligonucleotides, ensuring their integrity and facilitating their passage through the nuclear pore.
(another study):
Charge as a Selection Criterion for Translocation through the Nuclear Pore Complex
Colwell, Lucy J et al. “Charge as a selection criterion for translocation through the nuclear pore complex.” PLoS computational biology vol. 6,4 e1000747. 22 Apr. 2010, doi:10.1371/journal.pcbi.1000747
The first study still continues below
Liposome-oligonucleotide complexes are recognized more readily by nuclear import machinery due to their larger size and surface properties conferred by the liposomes.
So with plasmid DNA inside the LNP with the RNA, and the positively charged lipids inside, with DSPC, cholesterol, and peg, the net charge would be more negative on the LNP, and the zeta potential would also be much more negative.
This means the surface charge on the LNP is negative, even though lipids that are positive on the inside. But the nucleus cannot “see” those positively charged lipids INSIDE the LNP that is negatively charged (it has an outer negative charge, just like the liposomes in this study).
Negative charges on the liposomes facilitate their interaction with positively charged protamine, forming stable complexes that aid in the intracellular transport of the oligonucleotides.
”Protamine is polypeptide composed primarily of arginine. It has a molecular weight of approximately 4300 daltons and is highly positively charged.”
So not only, would negatively charged particles be attracted to the positively charged chromatin (yet another study showing negatively charged particles are driven through the nuclear pores due to charge), this study proved that negatively charged liposomes, facilitated the transport of the ODN right in through the nuclear pore to the nucleus (where the researchers wanted to change the genes).
It looks like gene mutations can occur when the LNP is negatively charged, enters through the nuclear pore, and zips right in.
The negatively charged liposomes (which can occur just the same with a lipid nanoparticle with a higher negative charge), are attracted to, the positively charged parts of the nuclear pore complex, and the chromatin inside the nucleus, and will act, as a nuclear localization signal to drive not only the contents of the liposome (which is that ring of lipids with ODN at the center of it) into the nucleus, but if the particle is small enough to enter, the particle itself, may enter the nuclear pore too (and interact with the contents, of the nucleus, of a cell).