Almost ALL biochemical reactions in the body involve movement of charged particles or the participation of charged molecules in some way.
This is a huge topic being ignored that is the driver behind almost all adverse events.
2/ There are positive (+) charges and negative (-) charges. A positive charge results from an excess of positively (+) charged particles--protons (+), within an object. A negative (-) charge is from an excess of negatively (-) charged particles--electrons, within an object.
3/ Positively charged particles are called "cations". They have more positive charges in them compared to negative. Negatively charged particles are called "anions". Charges can be be altered based on interactions with other objects (like a balloon!)
4/ Calculating charge on an object might look daunting, but it is simple math. This is remedial chemistry (even though taught later in organic).
You could look up videos on formal charge, valence shells, and the formula, but for simplicity, we are just going to use basic math.
5/ For calculating net charge, we add up the total number of cations and anions. Let's use table salt as an example. Salt is made up of sodium and chloride, existing in equal parts (1:1 ratio). Sodium (Na) has a charge of +1. Chloride has a charge of -1. If you add 1 + (-) you
6/ get ZERO (0). This means salt, table salt, is neutral. it has no net charge. 0 is neutral. The following is only for use of math, is not accurate in numbers, and is just for teaching. Imagine an LNP, and there are positive and negative charges inside of it. Let's say we have
7/ Five positively charged lipids (these are the charged ionizable lipids) inside an LNP, each with a +1 charge on them, and ONE piece of broken up modRNA, that had a negative charge of -5 (purely arbitrary numbers for teaching). We would add the five charged lipids plus -5, to
8/ get a total of zero. This mean LNP would have a neutral net charge. This is not zeta potential. Zeta potential tells us what the charge on the surface of the LNP is, in relation to the ions it is surrounded by, the pH, viscosity, etc.
Net charge:
9/ pH is a measure of how acidic or basic a solution is. The more protons (H+), which are positively charged in solution, the more acidic. The stomach is very acidic. Laundry detergent is a weak base. Acids have pH values less than 7, and bases have pH values greater than 7.
10/ pH is critical in chemical reactions, enzyme activity, and biological processes. Biological systems, such as blood and cellular environments, maintain a specific pH range for optimal functioning. Enzymes, which are essential for our functioning.
11/ When it comes to processes in the body, almost ALL processes are charge dependent. It makes no sense that zeta potential and charges are being ignored in conversations.
Types of charged reactions:
Coulomb's law:
Positive charges (e.g., protons) attract negative charges.
12/ Ion to Ion: Ions with opposite charges attract each other, forming ionic bonds. For example, in a salt crystal (sodium chloride, NaCl), positively charged sodium ions (Na+) are attracted to negatively charged chloride ions (Cl-).
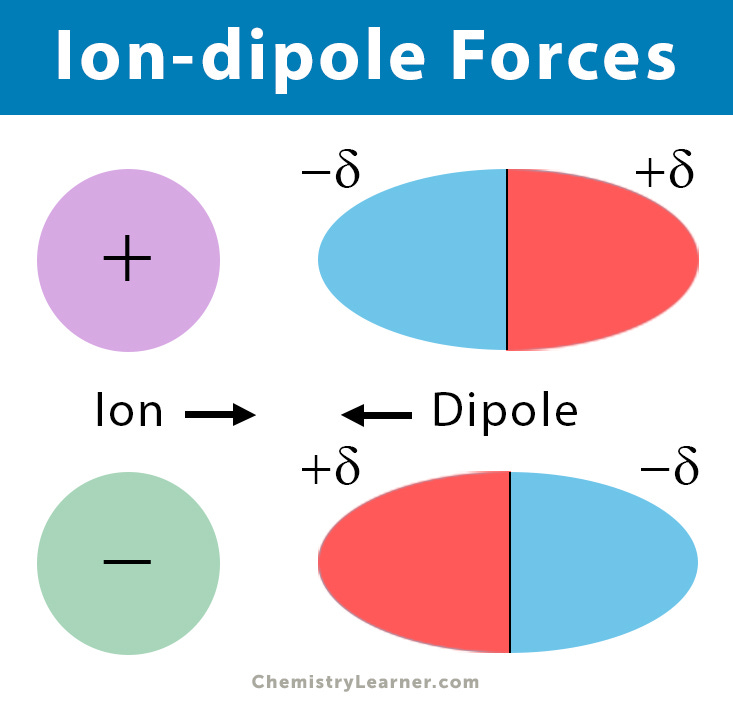
13/ Ion-Dipole Interactions:
Charged ions can interact with polar molecules, creating ion-dipole interactions. For example, in water, the positively charged hydrogen ions (H+) interact with the negatively charged oxygen atom (O) in the water molecule.
chemistrylearner.com/chemical-bonds…
Ion-dipole Forces (Interaction): Definition and ExamplesLearn ion-dipole forces, along with examples and diagrams. What are their characteristics. Also, learn ion-induced dipole forces.https://www.chemistrylearner.com/chemical-bonds/ion-dipole-forces
14/ Dipole-Dipole Interactions:
Polar molecules w/ partially positive and negative ends can interact through dipole-dipole interactions. The positive end of one molecule is attracted to the negative end of another molecule.
Polar molecules: distribution of charges is uneven:
15/ Hydrogen bonds occur between a hydrogen atom bonded to a highly electronegative atom (such as oxygen or nitrogen) and another electronegative atom in a different molecule (DNA).
Van der Waals forces :

16/ Electrostatic Shielding: IONS in the presence of other charges can partially screen the interaction between two charges. This means "screening" can MODIFY the interaction, through shielding or induction.
17/ For further reading, look up: electronegativity difference, dipole moments, intermolecular interactions. non-polar moments, and other electrostatic and bonding interactions between ions.
In the human body, some important charged interactions are: Nerve signaling: Action

18/potentials in nerve cells rely on the movement of charged ions (sodium, potassium, calcium, chloride) across cell membranes. Muscles: The sliding of protein filaments in muscle cells depends on the movement of ions (e.g., calcium) and ATP, which carries charges.
mitochondria:

19/involves charged ions (protons) creating an electrochemical gradient to produce ATP.
Immune cells use charged molecules and ions in signaling and recognition processes.
The body regulates charged ions like (Na+), (K+), (Ca2+), and Cl-) for maintaining homeostasis.

20/ Various cell signaling pathways involve the activation of proteins through phosphorylation and dephosphorylation, which are charge-based processes using phosphate groups (PO4³-) and protein kinases.

21/ Immune system: The interaction between immune cell receptors (e.g., T-cell receptors and B-cell receptors) and antigens is charge-based, involving electrostatic attractions and repulsions.
PHAGOCYTE: The negatively charged cell membrane of the phagocyte interacts with

22/ positively charged components on the pathogen's surface, facilitating the adherence of the pathogen to the immune cell.
Changes in ion concentrations can trigger immune cell activation and responses. Increase in intracellular calcium ions often signals immune cell activation.

23/ Mast cells, which are a type of immune cell, release histamine in response to allergens or pathogens. Histamine is a positively charged molecule that interacts with specific histamine receptors on nearby cells, leading to various immune responses, including inflammation.

24/ The charges on the surface on the LNP, and internally, can interact with an incredible number of biological processes, altering cell signaling pathways, cause clotting, protein coronas to form, immune system responses, cause acute hyponatremia, neurological concerns, and more