The Effects of Specific Antibiotics on Bifidobacterium--short and LONG TERM changes in the microbiome, especially on Bifidobacterium.
Antibiotics can significantly alter the microbiome, the community of microorganisms that inhabit various parts of the body, including the gut, skin, mouth, and other mucosal surfaces. While they can serve a purpose in times of need, they do come with adverse effects risks.
The microbiome can be altered by the introduction of antibiotics.
Reduction in Microbial Diversity
Antibiotics are designed to target and kill specific bacteria that cause infections. However, they can also affect non-targeted bacteria, leading to a reduction in microbial diversity. This reduction can be temporary or long-lasting, depending on multiple factors.
Selective Pressure
Antibiotics exert selective pressure on microbial populations, favoring the growth of antibiotic-resistant bacteria while suppressing susceptible strains. This can drive the numbers of antibiotic-resistant bacteria.
Disruption of Symbiotic Relationships
The microbiome consists of diverse microbial species that interact with each other and with the host in symbiotic relationships. Antibiotics can disrupt these relationships by altering the composition and abundance of microbial species.
Secondary Infections
Antibiotic treatment can create ecological niches within the microbiome that promote the overgrowth of opportunistic pathogens. This can increase the risk of secondary infections, such as Clostridium difficile infection, which is associated with significant morbidity and mortality.
Impact on Health
The microbiome plays a crucial role in various physiological processes, including digestion, immune function, and metabolism. Alterations in the microbiome due to antibiotic therapy can influence host health and contribute to the development of IBD, allergies, and other health concerns.
Resilience and Recovery
The microbiome does have the ability to recover over time. This process varies in individuals.
Bifidobacterium
Bifidobacterium is a genus of bacteria that belongs to the phylum Actinobacteria. These bacteria are Gram-positive, anaerobic, and typically rod-shaped. They are considered beneficial or "friendly" bacteria that inhabit the gastrointestinal tract, predominantly the colon, where they form a significant component of the gut microbiota.
There are several different strains of Bifidobacterium. These different strains will be explained in more detail in a later stack, alongside other important information.
Antibiotics have risks, and some directly impact the microbiome, especially Bifidobacteria.
Amoxicillin-Clavulanic
Amoxicillin-clavulanic acid, also known by its brand names such as Augmentin (among others), is a combination antibiotic medication used to treat various bacterial infections.
It contains two active ingredients:
Amoxicillin
Amoxicillin is a penicillin-type antibiotic that works by inhibiting the synthesis of bacterial cell walls, leading to bacterial cell death.
It is effective against a wide range of bacteria, both Gram-positive and Gram-negative, including Streptococcus pneumoniae, Haemophilus influenzae, Escherichia coli, and some strains of Staphylococcus aureus.
Clavulanic Acid
Clavulanic acid is a beta-lactamase inhibitor. Beta-lactamase enzymes are produced by certain bacteria to inactivate beta-lactam antibiotics, such as penicillins and cephalosporins, by breaking down their beta-lactam ring structure.
Clavulanic acid irreversibly binds to beta-lactamases, preventing them from deactivating amoxicillin. This allows amoxicillin to remain active against bacteria that produce beta-lactamases, extending its spectrum of activity.
Long-Term Changes in Human Colonic Bifidobacterium Populations Induced by a 5-Day Oral Amoxicillin-Clavulanic Acid Treatment
Study results:
Baseline Presence of Bifidobacteria
Before exposure to AMC, bifidobacteria were detected in all fecal microbiota samples from the 18 subjects (100%).
Short-term Impact
Following 5 days of AMC treatment, there was a significant decrease in the counts of total bifidobacteria. Some specific Bifidobacterium species, such as B. adolescentis, showed a notable reduction in occurrence compared to the reference period.
Long-term Impact
Although the total counts of bifidobacteria returned to baseline levels by day 8 after the end of AMC treatment, the diversity and composition of bifidobacterial species remained altered.
The average number of Bifidobacterium species per sample decreased significantly at day 5 compared to the reference period.
Some species, such as B. longum, remained stable after the antibiotherapy, while others, like B. adolescentis, showed a decreased occurrence.
Resilience and Resistance
While some individuals showed resistance to changes in their microbiota, with minimal alterations observed even during AMC exposure, others exhibited significant modifications.
The presence of intestinal beta-lactamases from individual microbiota could influence the response to AMC treatment.
Persistence of Changes
Even two months after the end of AMC treatment, the fecal microbiota did not fully return to baseline composition, suggesting long-term impacts of the antibiotic therapy.
Amoxicillin-clavulanic acid treatment can induce significant and persistent alterations in the abundance, diversity, and composition of bifidobacterial species within the gut microbiota of healthy individuals.
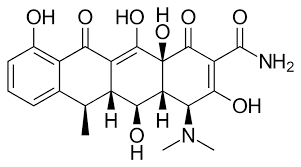
Doxycycline:
Doxycycline is a broad-spectrum antibiotic belonging to the tetracycline class. Tetracyclines are a group of antibiotics that inhibit bacterial protein synthesis by binding to the bacterial ribosome, thereby preventing the attachment of aminoacyl-tRNA to the mRNA-ribosome complex during translation.
Influence of oral doxycycline therapy on the diversity and antibiotic susceptibility of human intestinal bifidobacterial population
Study highlights:
Results:
Impact of Doxycycline Therapy
Doxycycline therapy led to a drastic reduction in the diversity and altered tetracycline susceptibility of intestinal Bifidobacterium populations.
Significance
The use of broad-spectrum antibiotics like doxycycline increased the prevalence of tetracycline-resistant commensal bacteria in the intestine.
Diversity
A significant decrease in the diversity of Bifidobacterium populations was observed during doxycycline therapy compared to the control group. (Average number of amplicons detected by PCR-DGGE: 0.8 in the antibiotic group vs 4.3 in the control group).
Antibiotic Resistance
Tetracycline Resistance: The proportion of tetracycline-resistant bifidobacterial population was higher in the antibiotic group (83%) compared to the control group (26%).
Gene Associated with Resistance
Resistance was associated with the presence of the tet(W) gene, detected through tet gene PCR.
Genetic Variability
Despite similar genetic fingerprints, different tetracycline susceptibilities were observed in strains from two subjects. A mutation in tet(W) causing lack of functionality was observed in one of the susceptible strains.
Azithromycin
Azithromycin is an antibiotic medication belonging to the macrolide class. It is used to treat a variety of bacterial infections caused by susceptible organisms.
Azithromycin works by inhibiting bacterial protein synthesis, thereby preventing the growth and reproduction of bacteria.
Broad Spectrum
Azithromycin has a broad spectrum of activity against many Gram-positive and Gram-negative bacteria, as well as atypical bacteria such as Mycoplasma, Chlamydia, and Legionella.Indications
Azithromycin is commonly prescribed to treat infections such as:Respiratory tract infections: Including bronchitis, pneumonia, and sinusitis.
Skin and soft tissue infections: Such as cellulitis and impetigo.
Sexually transmitted infections (STIs): Including chlamydia and gonorrhea.
Ear infections: Such as otitis media.
Certain gastrointestinal infections: Such as traveler's diarrhea caused by certain bacteria.
Prevention of bacterial endocarditis in dental procedures for patients with certain heart conditions.
Long Half-Life
Azithromycin has a long half-life, meaning it remains active in the body for an extended period after administration.Side Effects
Serious adverse effects are rare but can include allergic reactions and liver toxicity.Resistance
Overuse or misuse of azithromycin can lead to the development of antibiotic resistance in bacteria, reducing the effectiveness of this and other antibiotics in the future.
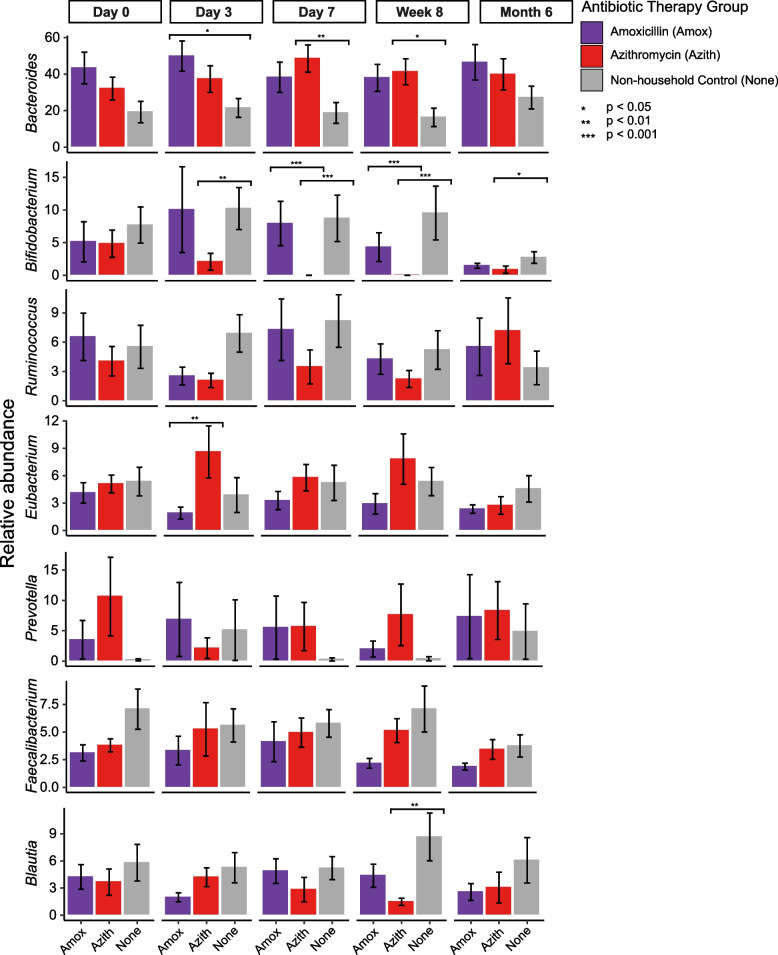
Common antibiotics, azithromycin and amoxicillin, affect gut metagenomics within a household
Comparison to Controls (Placebo or No Antibiotic Treatment):
After starting antibiotic therapy (after day 0), the relative abundance of Bifidobacteria in participants treated with azithromycin was significantly lower compared to controls.
Azithromycin treatment led to a reduction in the abundance of Bifidobacteria within the gut microbiota compared to individuals who did not receive antibiotics.
Comparison to Amoxicillin-Treated Participants:
At day 7 and week 8 of the study, the relative abundance of Bifidobacteria in participants treated with azithromycin was significantly lower compared to those treated with amoxicillin.
Azithromycin had a more pronounced effect on reducing the abundance of Bifidobacteria compared to amoxicillin at these time points.
“We performed a randomized, double-blind, placebo-controlled trial in a group of children aged 12–36 months, diagnosed with recurrent asthma-like symptoms from the COPSAC2010 cohort. Each acute asthma-like episode was randomized to a 3-day course of azithromycin oral solution of 10 mg/kg per day or placebo. Azithromycin reduced episode duration by half, which was the primary end-point and reported previously. The assessment of gut microbiota after treatment was the secondary end-point and reported in this study. Fecal samples were collected 14 days after randomization (N = 59, short-term) and again at age 4 years (N = 49, long-term, of whom N = 18 were placebo treated) and investigated by 16S rRNA gene amplicon sequencing.”
”Short-term, azithromycin caused a 23% reduction in observed richness and 13% reduction in Shannon diversity. Microbiota composition was shifted primarily in the Actinobacteria phylum, especially a reduction of abundance in the genus Bifidobacterium. Long-term (13–39 months after treatment), we did not observe any differences between the azithromycin and placebo recipients in their gut microbiota composition.”
Well, at least it resolved after a year.
There are other antibiotics which impact the gut microbiome, specifically, Bifidobacterium.
However, there is one debated topic involving a pathogen being implicated in the reduction of Bifidobacterium.
Correlation is not causation.
Until next time.